Lead (Pb) Natural Resources Locations In Nigeria And Its Applications

Nigeria is one of the Africa countries with abundant lead natural resources deposits and minerals.
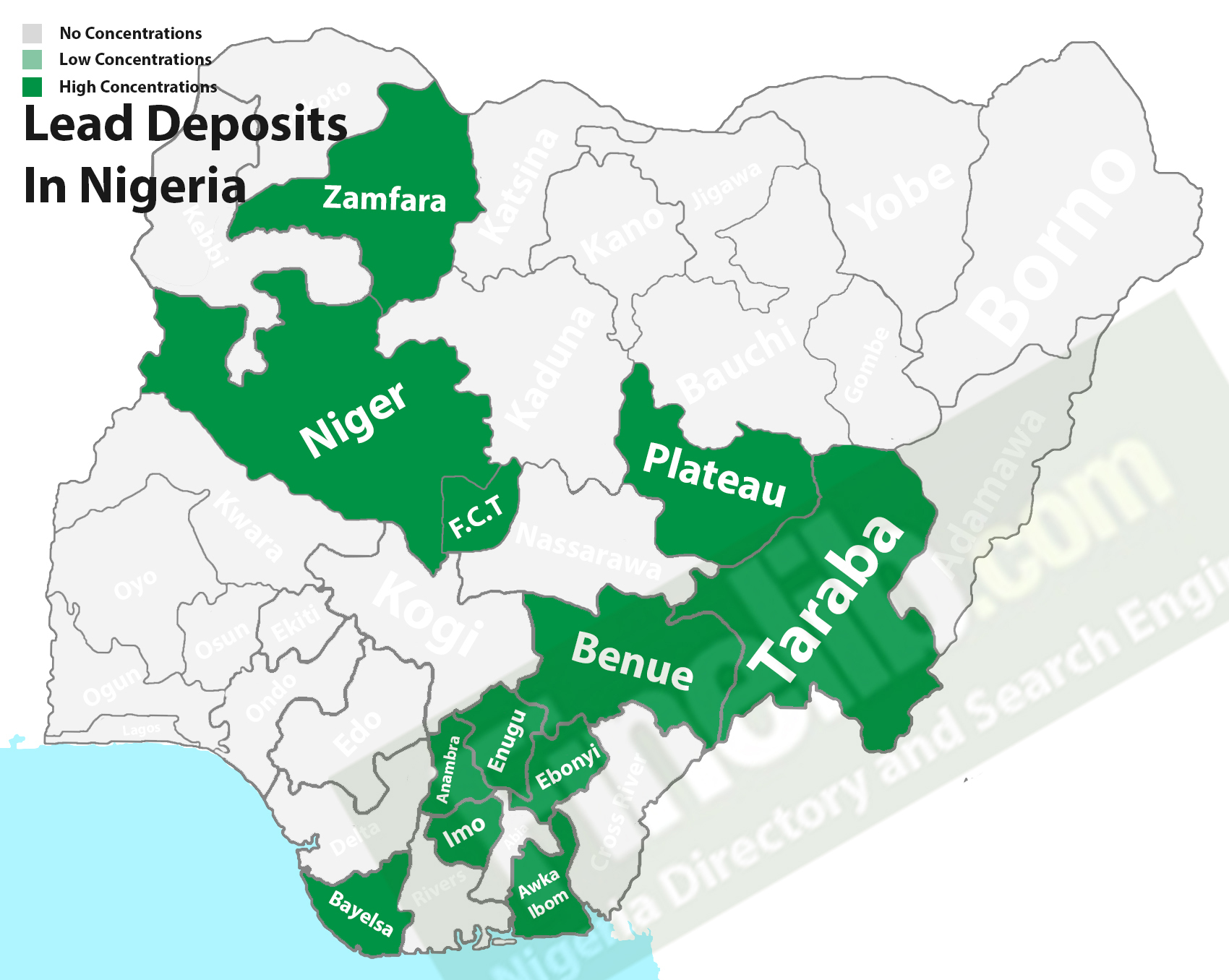
Listed below are some of the states in Nigeria that are rich in lead deposits, they are Akwa Ibom, Imo, Anambra, Bayelsa, Benue, Enugu, Niger, Ebonyi, FCT, Plateau, Cross River, Taraba, and Zamfara.
Lead is a metallic element primarily produced from the mineral, galena.
Most mineral ore contains lead with only about 3 of these minerals commercially viable, they are galena, cerussite, and anglesite and are mostly found in conjunction with other metals like zinc and silver.
Prices of Lead in Nigeria market
In Nigerian market, the current price of a ton of lead commodity is ₦1,654.30, which varies depending on location and sellers.
The global market for lead is driven by development. For this reason, the prices of this commodity range from ₦230,000 to ₦1,654.30 in Nigerian market.
Notwithstanding, the recycled lead scrap is always lower in price both locally and internationally.
The occurrence of Lead in Nigeria
The occurrence of lead in Nigeria added to the numerous abundant mineral resources in the country; though it has numerous applications which are quite useful in industries, it is generally also considered harmful when being exposed to.
There have been series of lead poisoning in Nigeria, West Africa through the intake of contaminated water especially in Zamfara state where series of death has occurred and this was attributed to underdeveloped domestic mining by the villagers which if not seriously taken into consideration by the government can even lead to the wiping away of an entire village.
Chemical Properties of Lead
Lead has an atomic number of 82, a melting point of 327.5 °C, and a boiling point of 1,749 °C. These are known as the chemical properties of lead minerals.
Physical properties
On the other hand, lead is very soft, blue-grey in color, has a high density, is ductile, malleable, resistant to corrosion, and has an electrical conductivity compared to other metals.
The mineral ores are mostly mined as a by-product of the above-stated valuable metals.
Ores of Lead
Extraction of lead minerals came as a result of three major ores, most times called the chief ores.
These are ores are dug from underground mines, and are considered the principal ores of lead.
Lead has three chief ores, which include:
• Galena - lead sulfide (PbS)
• Cerussite - lead carbonate (PbC03)
• Anglesite - lead sulfate (PbS04)
Apart from these 3 chief ores that are found in lead, other common lead-bearing minerals are:
• Boulangerite
• Minim, and
• Pyromorphite.
Which one is the best among the lead ores?
Galena is the best and most important ore of lead. It is the primary source of silver, commonly mined for its rich content.
Galena is a shiny grey, heavy metallic sulphide (PbS) from which lead ore is commonly found. It remains one of the widely distributed sulfide minerals, and the most abundant of all.
Today, this lead ore, galena, is mined at many locations across the globe, making up all of the ores mined presently.
Types of lead deposits
There are three major types of deposits from which lead is produced, they are:
• Sedimentary exhalative (SedEx) deposits
• Volcanogenic massive sulfide (VMS) deposits
• Mississippi Valley-type (MVT) deposits.
Sedimentary exhalative
Sedimentary exhalative, SedEx deposits are the major deposit that forms part of the total of the world's lead resources (up to 50%).
Formation of sedEx deposits
SedEx lead deposits are formed when hot liquids rich in metals are released in a water reservoir, usually the ocean.
The result thereafter becomes ore-bearing hydrothermal fluids, resulting from the precipitation from the final class of the hydrothermal deposit, stratiform ore at the ocean base, or the basin floor sediments.
Volcanogenic massive sulfide
Volcanogenic massive sulfide deposits are, in other words, known as VMS ore deposits.
These are the metal sulfide type of ore deposits which, for the most part, are lead and zinc. In addition to zinc, they contain silver, gold, and copper in significant amounts.
They are formed by volcanic-related hydrothermal processes in submarine settings.
Recently in modern oceans, VMS ores are the same as the black smokers precipitate formed on the seafloor around undersea volcanoes.
Mississippi valley deposits
Mississippi Valley-type (MVT) is the ore mineral replacement of carbonate-hosted sulphide rocks. They account for up to 25% of the world's lead and zinc resources and are found almost everywhere around the universe.
MVT deposits are strabound, epigenetic ore-bodies occurring mostly in the Mississippi valley region of the United States, chiefly composed of galena and sphalerite.
According to history, from the 19th century through the mid-20th century, MVT deposits were a key source of lead in the US.
Who Discovered Lead and Where
The existence of lead started about 7000 years ago, and it was in use by 3000BC.
The origin of the name "lead" arise from the Anglo-Saxon word for metal.
Lead is among the earliest metals founded by the human race, precisely extracted first by the ancient Egyptians.
The Egyptians and Chinese initially applied to lead many different forms such as in making sculptures and its compound in pottery glazes.
Around 2000BC, lead was used in China for the construction of forged coins, which dates back to the history of the Chinese currency.
The lead's property of resistance to corrosion was first discovered by the Greeks, and the resistance properties were further used on ship hulls as a protective covering.
In medieval times, the Romans started extracting lead in greater quantities for the construction of water pipes and bathtubs.
It was the Romans that gave 'lead' the name "plumbum", from which its chemical symbol 'Pb' was derived, and the popular modern word for "plumbing" was obtained.
Around the first century AD, Roman's production of lead increased drastically to a figure estimated at around 80,000 tons per year.
The use of lead as piping and corrosion protection continued from the first century through the 20th century.
In the middle ages, some parts of the European country started using lead as a roofing material due to its fire resistance property.
Westminster Abbey formally titled the Collegiate Church of Saint Peter at Westminster, and St. Paul's Cathedral London were roofed with lead roof hundreds of years ago.
In addition to the technical application of lead, the Romans used lead pots in an extensive range of recipe preparation processes such as grape crushing, and wines. According to them, they did so because they discovered overtones of the favorite flavor of lead that could add value to the wine.
The demand for lead materials kept on increasing continuously until the present time. And this actually makes the trading spread across continents across the globe.
Largest Producing Countries for Mined and Recycled Lead
Mined leads are produced in large quantities in the United States of America, China, and Australia.
Whereas the recycled lead is largely produced by the following countries: China, the USA, and Germany with China being the foremost lead producing country so far, thus, accounting for up to 60% of all lead production.
According to the US Geographical Survey (USGS), the largest ore deposits of lead can be found in countries like Peru 8%, Russia, United States of America 16%, China 18%, Canada 6%, and Australia 20%.
European countries, as a whole, are not recorded actually for having a large deposit of ore, but some counties within Europe such as Poland and Sweden have the biggest production values.
Prices of Lead Today
The price for 1 kilogram of lead commodity, which is equivalent to I ton (1,000 kilograms quantity) today is $1.65.
Although the price may not be static all through the year, the average price per ton of lead is 1.65 U.S dollars as of the 22nd of May, 2020. This price might change, as market demand changes.
Exporting countries
Countries that send lead ore commodities to another country for sale are the United State of America ($744M), Mexico ($1.22B), Peru ($1.1B), Australia ($345M), and Russia ($659M).
Countries Buying Lead
The top buying or importing countries for lead ore products according to the values they buy are:
China ($1.87B)
South Korea ($1.51B)
Germany ($460M)
Belgium-Luxembourg ($492M)
Canada ($544M)
Lead Ore Mining
Ores of lead are widely distributed across the globe. The mining of lead has become widespread to the extent that up to 44 countries around the world are now into lead mining.
The most commonly mined lead ore is the galena or lead sulfide, which makes up almost all of the ores of lead mined around the world today.
Among 44 countries that are into lead ore mining today, just about five (5) countries account for up to 68 percent of the total world's lead production.
Lead is usually found along with other minerals such as zinc, silver, copper, and gold (sometimes) during mining. This is so because large bulks of zinc deposits contain galena, which is lead ore.
Amongst the above-listed minerals which are mined alongside lead, zinc is found in great quantity and more often than others.
Different types of lead ores can be mined from the underground surface, but the three (3) ores of lead mined for lead production are galena, cerussite and anglesite.
According to the world's estimate, just about half of the lead produced globally is derived from mining activities. The remaining half is acquired from recycling processes, particularly car batteries.
Extracting Lead Materials from Its Ore
At this juncture, we shall the looking at how the lead is mined, not minding the location of the deposition.
Lead is extracted from ores dug from underground mines but for the extraction to be effective, lead ore has to be grounded into fine particles until it has a salt-like texture, after which it undergoes a flotation process, Flotation process means mixing the well-grinded lead ore with water.
In addition to the flotation process, pine oil and air bubbles are introduced causing agitation and making oil froth on the surface, the oil froth contains the lead which is scooped off and filtered to remove the water.
At about 2500oF – 3500oF, the powder is sintered to oxidize impurities like sulphur and is heated further in a blast furnace, this makes the carbon produce molten Lead which flows into lead moulds, to be 99% pure it is further subjected to melting and skimming.
Since is found in conjunction with other metals such as gold and silver, the precious metals can be removed by adding to it a small quantity of zinc.
The gold and silver dissolve more easily in zinc than in lead, and when the bullion is cooled slightly, zinc dross rises to the top, bringing the other metals with it.
Extraction of lead ore from the underground surface is achieved by the method of rock drilling. This is done using the heavy-duty machinery of blasting with dynamites.
The extracted lead ore is further transferred for processing, during which the ore will, first of all, undergo concentration.
At this stage of concentration, the ore is been set apart from the waste rock. Then the next step would be grinding into smaller particles.
Lead manufacturing or preparation
Lead preparation, in other words, called the making of lead, is a complex process involving a number of steps. It starts with melting the ground particles of lead ore.
The ore is melted at over 300 degrees Celsius. Meanwhile, a mix of sand is shoveled onto a long casting bench, where it is turned and mixed by hand to create an even consistency.
At the discretion of the caster, moisture is sprayed onto the sand, which will allow the molten lead to form evenly.
Next, level out the sand flat, float the sand carefully, and smooth. The sand which prevents the molten lead from forming a rough, uneven surface with the lead melted, any dross impurities can be removed.
Carry out another quick final spray to seal the sand for the pour. At this point, position a screed rail, ready to smear the liquid metal.
With all impurities removed, the molten lead is ready for pouring. First, drain up to 92% into a hydraulically, powered ceramics trough. Then, at a constant rate, the trough is mechanically tipped.
Pour the melted ore, and use the screed rail to smear the molten lead across the casting bench. Seconds later, a solid lead sheet is formed, and then you can measure, cut, and trim to the desired specification.
Applications of lead
• Lead is used in building construction such as roofing material, cladding, flashing, roof parapets, gutters, and gutter joints.
• Used in lead-acid batteries, bullets, pewters, fusible alloys, and as a radiation shield.
• It’s used in Water pipes.
• It’s used for the ballast keel of sailboats due to its high density and resistance to corrosion.
• Lead is added to brass to reduce machine tool wear.
• Lead is used as electrodes in the process of electrolysis.
• It is used in solder for electronics.
Final words
Lead and its ores have been known for a long time, however, have only been mined past on a very small scale. Today, lead occurs in very large quantities in different parts of the state across the country.
And presently, due to the increasing demand in the market, the estimated reserve lead for trading in Nigeria is well over 10 million tons.




